Organic Chemistry Basic Principles
Catenation
Chain forming, branch forming, ring forming tendency of carbon
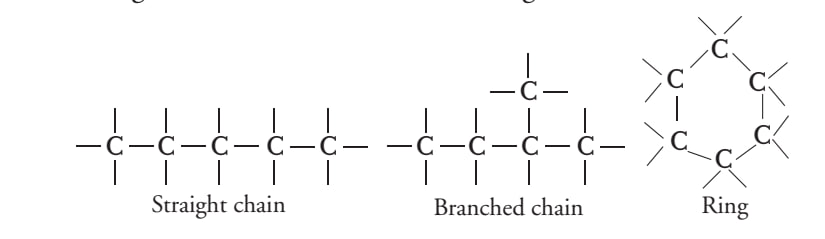
Tendency to form multiple bonds
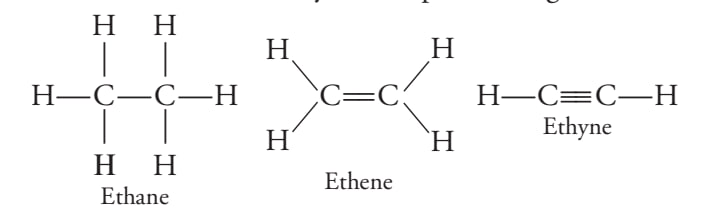
C-C bond - saturated hydrocarbons
C=C , C=C - unsaturated
Carbon can exhibit isomerism
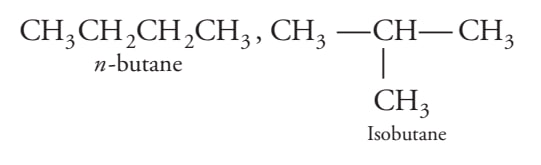
iso - same, mer = molecule
Structural representations of organic compounds
electron dot or dash structure
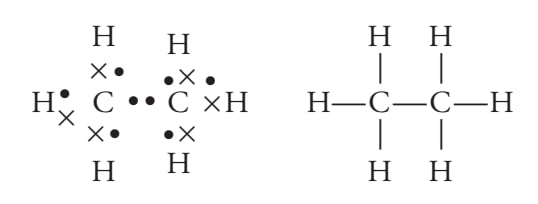
Condensed structure
Bond line structure
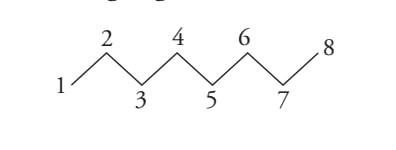
CH3(CH2)6CH3
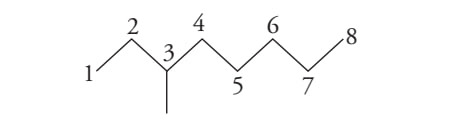
3-methyl octane
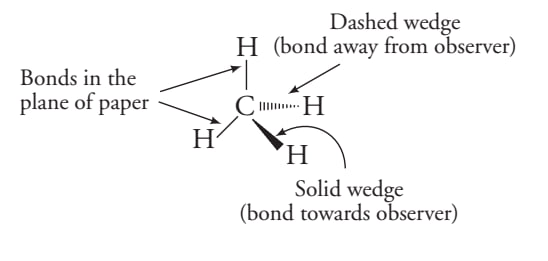
Three dimensional structure
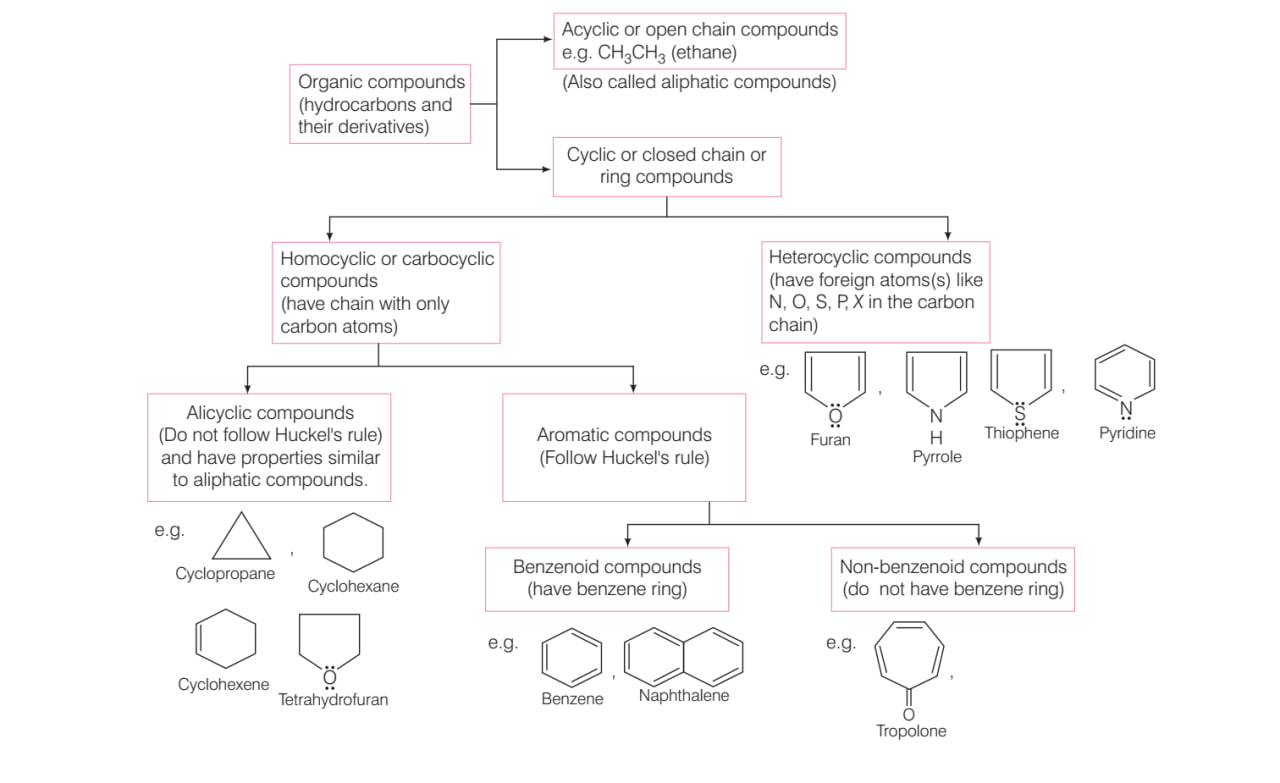
CLASSIFICATION OF ORGANIC COMPOUNDS BASED ON CARBON SKELETON
Voice message 08:38
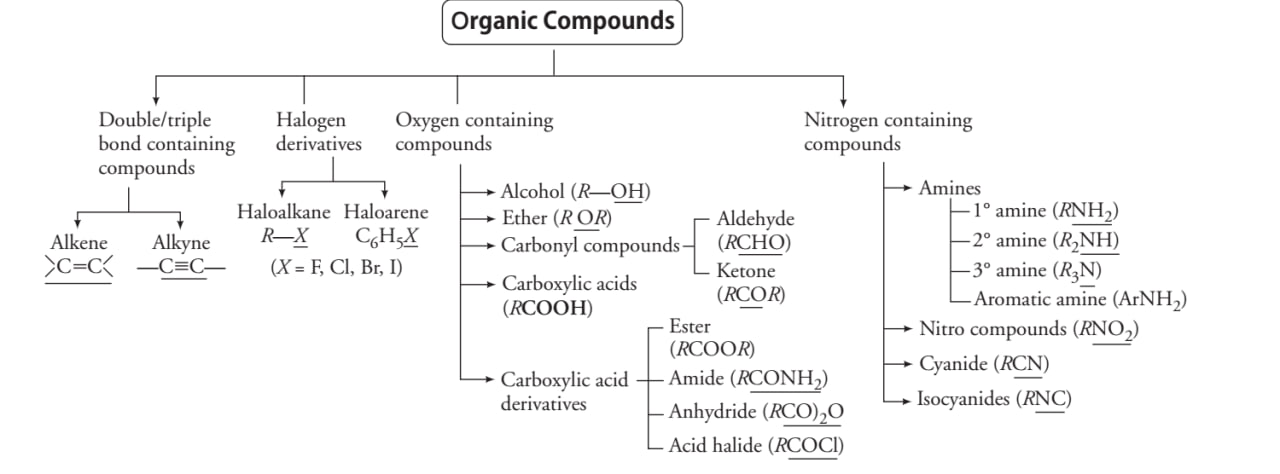
CLASSIFICATION OF ORGANIC COMPOUNDS BASED ON FUNCTIONAL GROUPS

Voice message 02:58

Voice message 02:11

Voice message 02:49
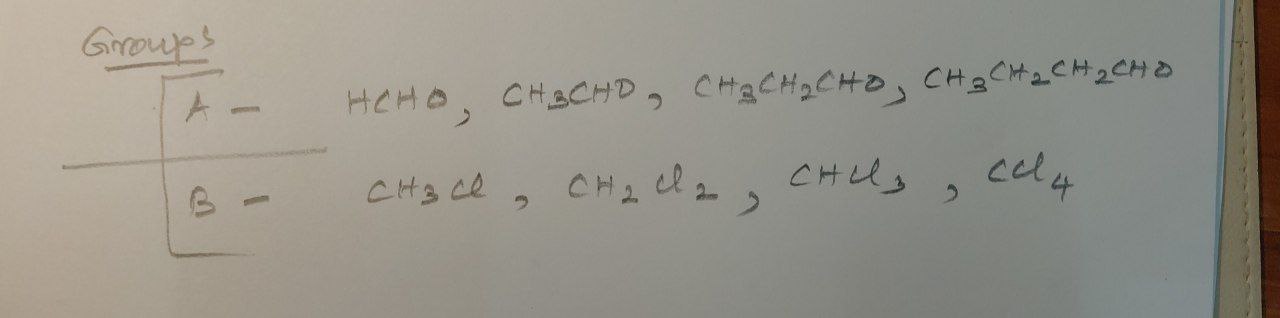
Which group is homologous? What is the molecular mass difference in the homologous series
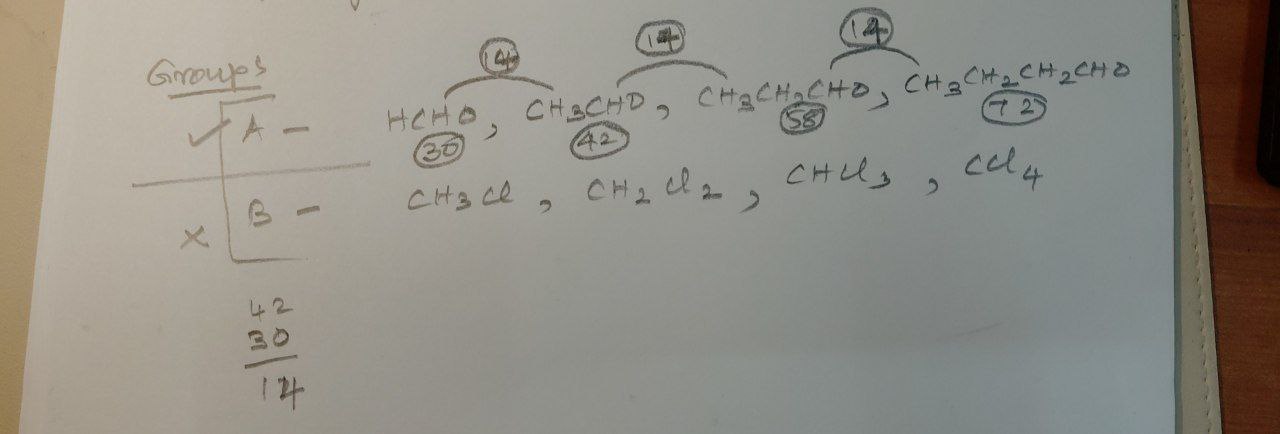


Voice message 06:04
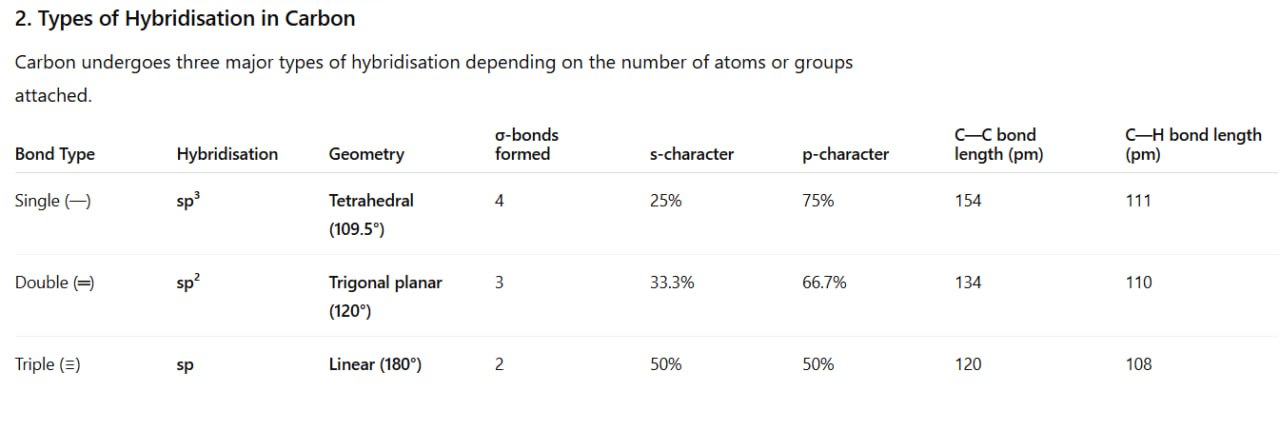
Hybridisation in Organic Compounds
1. Concept of Hybridisation
Hybridisation is the process of mixing atomic orbitals of nearly the same energy to form new equivalent orbitals known as hybrid orbitals.
These hybrid orbitals form σ-bonds and determine the shape and geometry of organic molecules.

Voice message 05:47
Voice message 00:53